
|
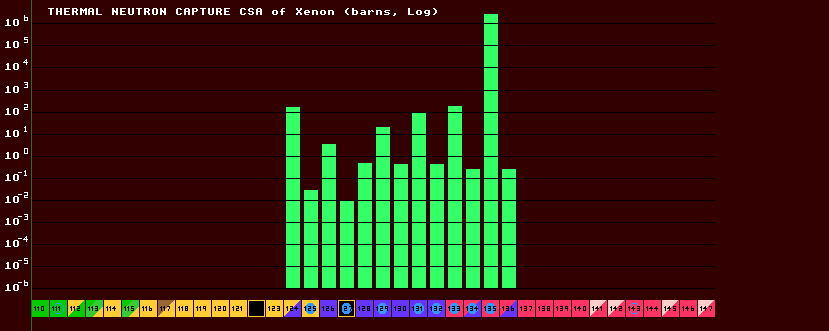
Slow moving neutrons (thermal neutrons, those with energies of order 0.05eV) are easily captured by nuclei, which may then become neutron rich and decay by The above chart shows the thermal neutron capture cross sectional areas for some stable and un-stable isotopes of xenon. The thermal neutron csa for Xe-135, the nuclear reactor poison, is extremely high; being four magnitudes higher than any other isotope of xenon. Note also the very distinctive odd/even 'comb-shaped' bars - the isotopes with an odd number of neutrons (odd-N) have a higher csa for thermal neutrons, they would like to capture a neutron to become an isotope with an even number of neutrons, and if they do so, their apetite for capturing another neutron is much diminished (much lower thermal neutron csa for even-N nuclei). Note that xenon, Z=54, has an even number of protons (Z-even), so xenon nuclei with N-even also have an even number of alpha particles (Helium nuclei) within them. Nuclei like to possess an even number of helium nuclei (N-even Z-even), for then they are more stable. See Nuclear Spin for the reason.
|
![]()