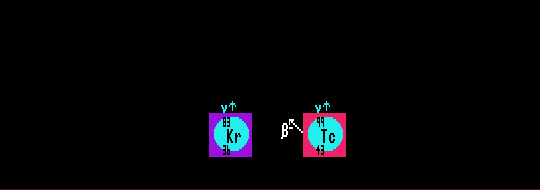
ISOMERIC TRANSITION
ISOMERIC TRANSITION (I.T.) or INTERNAL CONVERSION
A nucleus usually exists in its ground state, where individual nucleons pair up as best they can subject to shell constraints into a minimum total spin (which may not be zero and can be as high as 11/2) (the total spin of Even-N even-Z nuclei in their ground state is always zero). But in some nuclides, especially those around magic shell numbers (islands of isomerism), one or more nucleons can be excited into higher spin states. For some nuclei there are many such higher spin states. The most probable way for a nucleus to find itself in a higher spin state is just after decaying by more conventional means, such as by alpha decay, from where it can revert back to the ground state spin with the emission of gamma radiation, which is usually shed immediately. But if this emission is delayed by more than 1 microsecond, then it is said to be a nuclear isomer, and the process of releasing the excess energy is called an Isomeric Transition or Internal Conversion.
Unlike all other radioactive decays, isomeric transition does not involve the transmutation of one element into another element, it merely loses excess spin by emitting gamma radiation. This isomeric transition has an associated halflife, separate from any other halflife related to the isotope.
In the Segre chart, nuclides that can exist as isomers are shown by a cyan circle and the associated isomeric transition halflife in cyan coloured numbers. Even otherwise stable nuclides can exist as nuclear isomers. Isomers of heavy nuclides which find they are liable to undergo spontaneous fission instead of (or as well as) internal transition are called 'fertile isomers'.
Shown is stable krypton-36 which can exist as an isomer with a halflife of 1.86 hours and technetium-99, a beta decayer with a halflife of 213,000 years, which can also exist as an isomer with a halflife of 6.01 hours.
See Fertile Isotopes. and Nuclear Spin

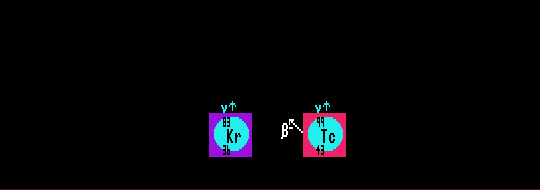
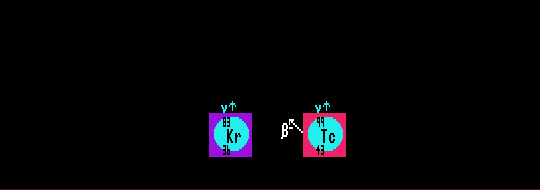
![]()