
|
98 CALIFORNIUM Cf (California)
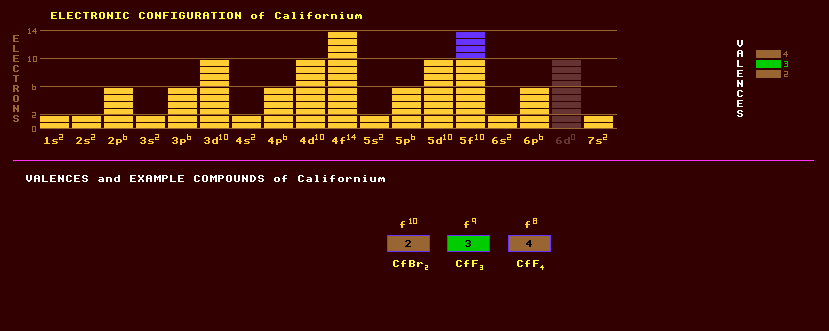
An artificially produced, highly unstable, transuranic rare earth element of the actinide series with no detectable natural occurrence on Earth. Californium was first produced in 1950 by bombarding microgram quantities of curium-242 with 35MeV helium-4 ions in the Berkeley 60 inch cyclotron in America resulting in the isotope californium-246 which decays by either alpha decay or spontaneous fission with a halflife of 1.5 hours. Californium-249, which is an alpha emitter with a halflife of 351 years, is produced by the beta decay of berkelium-249, which has a halflife of 320 days. The heavier isotopes of californium are produced by irradiating berkelium-249 with neutrons, which becomes berkelium-250, a beta/alpha emitter with a halflife of 3.217 hours, some of which then decays by the beta decay route to become californium-250, which absorbs more neutrons to become progressively heavier: first californium-251 then californium-252, etc. Californium-250 decays by either alpha or spontaneous fission with a halflife of 13.1 years; californium-251 by alpha decay with a 900 year halflife; and californium-252 by alpha or SF decay with a 2.64 year halflife. Californium-252 is a very strong neutron emitter (the SF route) and represents a biological hazard, but finds use in neutron moisture gauges, in oil well exploration, in cancer treatment by radiation therapy, and as a portable neutron source for on-the-spot gold and silver activation analysis in prospecting. Californium expresses a valency of +4 or, more usually, +3. Californium trichloride, CfCl3, californium oxychloride, CfOCl, and californium oxide, Cf2O3 were the first compounds of californium to be prepared. Of the halides, the dibromide, CfBr2, and diiodide, CfI2, the trifluoride, CfF3 and trichloride, CfCl3, and tetrafluoride, CfF4 have been prepared. Of the oxides, the sesquioxide, Cf2O3, and dioxide, CfO2 are known and a monoxide, CfO, may exist. A [Cf(C5H5)3] complex is also known. The longest lived isotope of californium is californium-251 with a halflife of 890 years decaying by alpha decay into the alpha decaying curium-247 which has a halflife of 15.6 Million years. Altogether, 18 isotopes of californium are known, all radioactive, and ranging from the alpha decaying californium-239 which has a halflife of 42 seconds to californium-256 which decays by spontaneous fission. Claim to fame:
|
![]()