
|
59 PRASEODYMIUM Pr (Greek: prasios didymos = green twig)
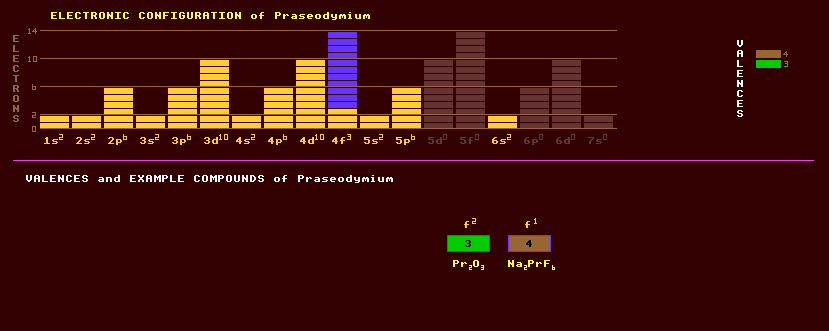
A soft, malleable, ductile silvery white rare earth metallic element of the lanthanide series. It is somewhat more resistant to corrosion in air than europium, lanthanum, cerium, or neodymium, but when exposed to air develops a green oxide that spalls off. As with other rare earth metals, it should be kept under a light mineral oil or sealed in plastic. Misch metal, used as a pyrophoric alloy in cigarette lighter flints, contains about 5% praseodymium, the rest being cerium, lanthanum and neodymium. Praseodymium, like all lanthanides rare earths, is predominantly trivalent, but also forms tetravalent compounds. The oxide is yellowish-green, the salts green. As with other rare earth, compounds of these elements in solution have distinctive sharp spectral absorption bands or lines, some of which are only a few Angstroms wide. Salts of praseodymium are used to colour enamel and mixed with other materials give a particularly intense yellow coloration to glass. The rare earth oxides, including Pr2O3, are among the most refractory substances known, along with other rare earths, it is widely used as a core material for carbon arcs used by the cinema industry for studio lighting and projection. Salts of praseodymium are used to colour glasses and enamels; when mixed with certain other materials, praseodymium produces an intense and unusually clean yellow colour in glass. Didymium, once thought to be an element but now known to be a mixture of neodymium and praseodymium, is a colourant for welders goggles. The element occurs along with other rare earth elements in a variety of minerals, of which monazite, (Ce,La,Nd,Th)PO4 and bastanite are two principal commercial sources. Praseodymium exists as just one stable isotope, praseodymium-141. Altogether 28 radioactive isotopes are known, ranging from the inverse beta decaying praseodymium-124 the the beta decaying praseodymium-154. Claim to fame:
|
![]()