
|
5 BORON B (Arabic: buraq)
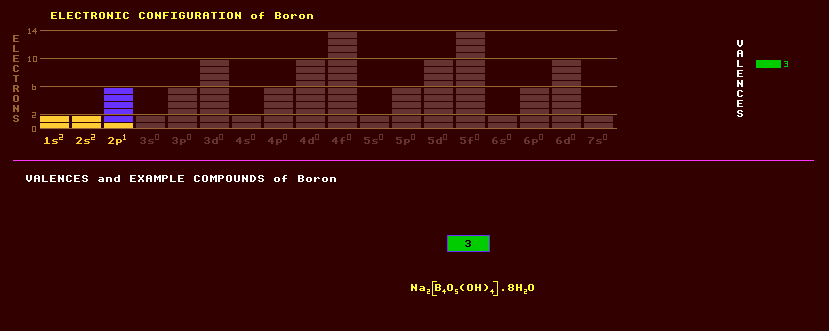
Boron is an amorphous yellowish-brown element which can be formed into a conducting metal. Boron is of utmost importance in nuclear reactors, because of its great absorption cross-section for neutrons; thus boron steel is used for the control rods. Boron turns flames green and is used in pyrotechnic flares. Boron is used as an igniter in chemical rockets. High purity boron is produced by the electrolysis of molten potassium fluroborate and potassium chloride. Boron is essential for plants for reasons unknown. Boron can exist in a multiplicity of different polymorphic forms, based on a great variety of three dimensional structures of interconnected boron atoms. These structures include the regular icosahedron, a 20-sided platonic solid; the truncated icosahedron, a football-like structure of hexagons and pentagons; and the truncated tetrahedron. These three basic shapes are interconnected or condensed to form much larger geodesic structures, comprising of as many as 192 boron atoms. The nearly spherical B84 consists of an inner B12 icosahedral shell bound by another 12 bridging boron atoms to an outer truncated icosehedron shell, B60, which has the same structure as Buckminsterfullerene, C60. Boron is of low abundance but found in nature as borax, hydrated sodium borate, Na2B4O7·10H2O. Boron has similar chemistry to silicon, although belongs to group 13 rather than group 14, but borates are rather more soluble in water than are silicates; borax occurs as salt precipitates from hot springs in volcanic areas. Borax beads are used in chemistry to help determine an unknown metal from the colour of the glassy bead formed in a bunsen flame. Borax is used as a flux in welding. Boric or boracic acid, H3BO3, is used as a mild antiseptic. It loses water on heating to form metaboric acid, H2B2O4, which on further heating forms tetraboric acid, H2B4O7. Heating this to higher temperatures results in boric oxide, B2O3, an amphoteric oxide with both acidic and basic properties. Boron compounds are used as enamels on domestic white goods. Boron forms a complex series of hydrides called boranes, but unlike as in hydrocarbons where the carbon is tetravalent, boron is trivalent, thus compounds like borane BH3, diborane B2H6, etc. form, some are used as rocket fuels. Many boron compounds are also cage like with three dimensional structures: thus B4Cl4 is tetrahedral with four triangular faces, B8Cl8 is dodecadeltahedral with 12 triangular faces, B9Cl9 is a tricapped trigonal prism with 14 triangular faces, [B6H6]2- is octahedral with eight triangular faces, [B7H7]2- is pentagonal bipyramidal with ten triangular faces, [B10H10]2- is bicapped square anti-prismatic with sixteen triangular faces, and [B12H12]2- is icosahedral with twenty triangular faces. Boron can be combined with a metal producing borides, some of which are very hard and heat resistant. Boron can be used to dope silicon to make a p-type semiconductor, used in transistors. Boron is used in sodium borosilicate glass, or pyrex, having a low thermal expansion coefficient and high chemical resistance. Boron carbide, B4C, is very hard and used for cutting tools. Boron nitride or Borazon, BN, has the same crystal structure as diamond but is harder than diamond and exhibits the unusual property of being electrically insulating yet conducting heat like a metal, and has lubricating properties like graphite. It appears that almost all of the Earths' boron (and lithium & beryllium) was created by extra-terrestrial cosmic ray bombardment of heavier nuclei in the interstellar medium causing their fragmentation in spallation reactions before the formation of the solar system. Kernite, Na2B4O7·4H2O, another hydrated sodium borate but with less water of crystallization than borax, is the principle ore of boron. Borax or tincal, yet another hydrated sodium borate with six more water molecules, Na2[B4O5(OH)4]·8H2O, is a soft, white earthy mineral found in salt pans and solar evaporite deposits used in the making of boric acid and also as an ant poison in the home. Ulexite, otherwise known as boronatrocalcite, NaCa[B5O6(OH)6]·5H2O a hydrated sodium calcium borate, is an extremely soft clear/white mineral ore of boron found as an evaporite deposit. Colemanite, hydrated calcium borate, Ca[B3O4(OH)3]·H2O, is one of the main ores of boron. Boracite, Mg[ClB7O13], is used in the production of borates and of boric acid or boracic acid which is used as an eye bath in the home. Danburite, calcium borosilicate, CaB2Si2O8, is of interest only to scientists and collectors. Datolite, hydrous calcium borosilicate, CaBSiO4(OH) is just a minor ore of boron. The prismatic blue or violet dumortierite, aluminium borosilicate, Al7(BO3)(SiO4)O3 is used to make aluminous refractories. The precious gemstone tourmaline is a complex borosilicate of varying composition, Na(Mg,Fe,Li,Mn,Al)3Al6(BO3)3(SiO3)6(OH,F)4, with the most multifarious colours of any gemstone and occurs as seven distinct species: elbaite (multi-hued), schorl (black), buergerite and dravite (brown), rubellite (coloured pink by lithium), chromdravite (green), and uvite (black, brown or yellow-green). There is also a green verdelite and a blue indigolite. The colourless achroite is the rarest. Besides being strongly pleochroic, that is displaying different colours from different directions, tourmaline crystals are often bi-coloured, being perhaps pink at one end and green at the other, and may be up to a metre in length. Tourmaline is strongly pyroelectric, and generates sufficient static electricity (positive at one end and negative at the other) by the heat of the hand to attract wood ash. Rubellites (pink) and verdelites (green) are the most popular because of their colour. Tourmaline is used in optics for the polarisation of light. Boron consists of two stable isotopes, Boron-10 at 20% and Boron-11 at 80% isotopic composition. Boron-10 is used to detect neutrons, as when it absorbs one, it breaks into two easily detected charged particles, lithium-7 and helium-4. Boron-10 is produced in the atmosphere by cosmic ray bombardment. Boron compounds are cumulative poisons, borax is used as ant powder. Claim to fame:
|
![]()