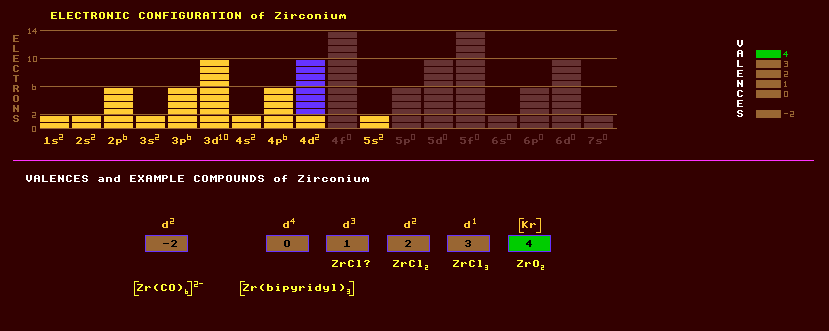

|
40 ZIRCONIUM Zr (Arabic: zargun = gold colour)
Zirconium is a hard grayish-white lustrous metal which when finely divided may ignite spontaneously at elevated temperatures. Like magnesium, the solid is much less ignitible. Zirconium wire is used in photographic flash bulbs where is is electrically ignited and burns in a brief intense white flash, it is also used as a getter of oxygen in vacuum tubes. Zirconium, when purified of hafnium contamination and fashioned into zircalloy tubing, is used to contain nuclear fuel rods because its absorption of neutrons is very small and it is extremely resistant to the highly corrosive environment of a reactor. Zirconium is exceptionally resistant to many common acids and alkalis and sea water, thus it finds use in the chemical industry. Zirconium is considered to be of low toxicity. Alloyed with niobium, zirconium becomes superconducting at low temperatures and is used to make electro-magnets. Alloyed with zinc it becomes magnetic at 35 Kelvin. The oxide, zirconia, or zirconium dioxide, ZrO2, is white and is used as a pigment and as a refractory material in jet engines and nuclear reactors. Some laboratory crucibles are made of zirconia. Man-made pure crystalline zirconia is colourless with a high index of refraction like diamond (the index of refraction of diamond is 2.42), and when cut is used for imitation diamond in jewellery. Zirconium dioxide is used as one of the anti-reflective coatings on multi-coated camera lenses etc. Moon rocks contain more zirconium (as oxide) that do terrestrial rocks. Zirconium is abundant in S-type stars and has been identified in the sun and meteorites. Zirconium carbonate is used as the antidote to poison ivy as it combines with urushiol, the poisonous constituent of poison ivy.
The ceramic crystal zirconium tungstate, Zr(WO4)2, (which contains ZrO6 octahedra and WO4 tetrahedra) is unique in being the only known material which has a negative temperature coefficient of volume expansion, that is, it contracts in all directions on heating over a very large temperature range. Some other materials have a negative expansion coefficient in one direction but a positive tc in the other directions. Most materials that exhibit negative thermal expansion only exhibit it over a narrow range of temperature apart from zirconium tungstate where the negative expansion occurs over a very wide temperature range: from near absolute zero to 800 Celsius. The contraction of zirconium tungstate amounts to 0.75% on heating from 1 Kelvin (near Absolute Zero) to 777 Celsius, when it decomposes into the oxides ZrO2 and WO3. Moreover, the magnitude of the effect is greater than that for most materials with a positive thermal expansion coefficient. The reduction in volume with increasw in temperature occurs because the oxygen atom in the Zr-O-Zr bond librates up and down with increasing temperature, effectively shortening the Zr-O-Zr bonds, bringing the zirconium atoms closer together, thus increasing the density of the zirconium tungstate. It's value lies in the possibility of obtaining a ceramic with zero temperature coefficient of expansion by mixing it with other ceramics, which would be of great use for constructing telescopes, pendulums, etc. Other substances change in volume on undergoing a phase change - see Crystalline Form.
Many zirconate crystals are used as ultrasonic transducers, being strongly piezo-electric. Some zirconates (and many niobates, tantalates and titanates) crystallize in the anisotropic perovskite structure and generally exhibit a range of active phenomena. Thus lead zirconate, PbZrO3, is both piezoelectric and anti-ferroelectric with a Curie temperature of 233 Celsius, see lithium niobate under niobium. Lead zirconate - lead tantalate solid solutions (PZT crystals) have ferroelectric, anti-ferroelectric and paraelectric phases, depending upon the proportions of the two components, and are widely used as piezoelectric and electro-optic crystals. The structure of each cell in PZT crystals can be switched between two different bistable states by an electric field, and this is the basis of some new ferroelectric random-access memories, or FRAMs, which can be made much smaller than the semiconductor capacitor based RAMs. The very hard and heavy mineral zircon, zirconium silicate, ZrSiO4, an important ore of zirconium, crystallizes in the tetragonal system and occurs in three polymorphic forms differing in density and optical properties. Zircon is an important ore of zirconium, and may contain up to 20% hafnium when it is known as hafnon. Zircon can be yellow, red, or brown (hyacinth); blue (starlight); gray-green; opaque; or pale yellow to colourless (jargon). Zircon is quite often weakly radioactive, with up to 4% of the zirconium being substituted by thorium or uranium, which destroys its crystalline form (metamict). Some varieties of zircon (those with radioactive elements) are thermoluminescent and will glow intensely for a moment (once only) when heated. Cyrtolite is a radioactive zircon containing yttrium and uranium. Heating may also restore the crystal structure of the metamict (radiation-damaged) varieties, and can also change the colour of zircons. Starlight, the clear sky-blue zircon variety used in jewellery, is made by heating the opaque brown stones in an oxygen-free atmosphere. Heating starlight zircon in air will then produce the golden variety, but both processes produce some colourless stones. All heat-induced colours can revert back to the original colour, a process hastened by sunlight, but may be restored again by careful heating. Zircon, like calcite, is strongly birefringent (with two indices of refraction, 1.94 and 2.02). The clear and coloured varieties of zircon are prized as gemstones, the radioactive varieties used as ores of uranium. The oldest rocks ever found on Earth are zircons dated at 4.2 thousand million years. Natural zirconium occurs as a mixture of four stable isotopes, 51% Zr-90 with lesser amounts of Zr-91, Zr-92, Zr-94; and 3% of Zr-96 which is radioactive with an enormously long halflife of 3.6 1017 years decaying by beta decay. Altogether, twenty radioactive isotopes are known. Claim to fame:
|
![]()