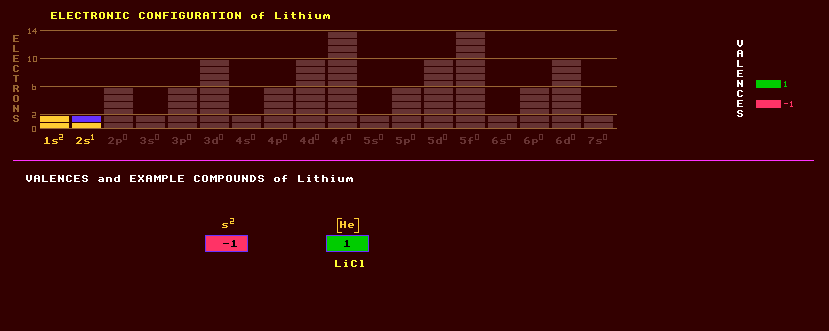

|
3 LITHIUM Li (Greek: lithos = stone)
Lithium is the least dense solid, relative density 0.585, an alkali metal chemically resembling sodium, but less active. It is a soft, white silvery metal, the surface of which tarnishes quickly when exposed to air. Present in tiny amounts in sea water, it is used in alloys to impart lightness and in the production of tritium; also as a basis for lubricant grease which have a high resistance to moisture and extremes of temperature, and as an ingredient of high-energy fuels. It is obtained by the electrolysis of molten lithium chloride. An exotic alloy of lithium and aluminium is also finding strategic use in military aeronautics and spacecraft because of its lightness coupled with great strength, being 38% lighter than aluminium. Lithium is also alloyed with magnesium for similar reasons. Lithium finds use in high-energy batteries known as Lithium batteries, where the high electropositive nature of lithium contributes to the very high e.m.f. (about 3.6 volts) obtained with lithium cells. Lithium batteries can have very long shelf lives of up to 10 years, though 5 is more common. The batteries have the highest energy density of any chemical battery, but cannot deliver high currents. They are based on a cathode of lithium cobalt iron oxide, a carbon anode and an electroluyte. Lithium-ion rechargeable batteries have a lithium cobalt oxide cathode and a carbon anode. They have a very high energy density, but charging and discharge currents can only be relatively small, and the battery, although better at retaining charge than nickel cadmium or nickel metal-hydride batteries, the charge does leak away much faster than it does in (non-rechargeable) alkaline batteries. Another disadvantage is that it must not be allowed to fully discharge, but, conversely, keeping a fully charged battery at high temperatures (over 20 Celsius) reduces its overall capacity over time. However, if nano-crystals of lithium tantalate coat the surface of the anode (which increases the surface area 30 fold), much higher charging and discharge rates can be achieved, sufficiently to fully charge the battery in 3 minutes flat. Lithium salts colour a flame crimson. It appears that almost all of Earths lithium (and beryllium & boron) was created by extra-terrestrial cosmic ray bombardment and fragmentation of heavier nuclei in the interstellar medium before solar system formation. The main ores of lithium are: spodumene LiAlSi2O6 a lithium aluminium silicate of the pyroxene group. Spodumene is a transparent trichoic mineral changing colour when viewed from different angles. Lepidolite KLiAl(F,OH)Si4O10 a phyllosilicate of the mica group with a pinky lilac colour, pentalite LiAlSi4O10 and amblygonite LiAl(F,OH)PO4 a hydrous lithium aluminium phosphate. Lithium is in-essential for life, but is used medicinally in the treatment of manic-depressives. As the deuteride, lithium deuteride, 6LiD, it is used inside hydrogen bombs to hold the element to undergo fusion, deuterium, in solid form, and also combined with tritium as 6LiT. It performs a second function within hydrogen bombs, and that is when the lithium-6 is bombarded by neutrons, it breaks up forming more fusible tritium. Only two stable isotopes of lithium are known, lithium-6 constituting 7% and lithium-7 present at 93%. A further four radioactive isotopes are known, ranging from lithium-5, which has an incredibly short halflife of only 3×10-22 seconds and decays almost instantaneously into a proton and an alpha particle, to lithium-11 which has a halflife of 9 milliseconds and decays by one of several routes emitting a combination of electrons, neutrons or alpha particles. Lithium was created in the Big Bang formation of the Universe, when Hydrogen and Helium were created too. The predictions of the amounts of Hydrogen and Helium formed at the same time is nearly correct, but the predicted amounts of Lithium formed then fall short; about 2/3rds of the Lithium that should have been produced is missing and no one can explain why. It is known as the 'cosmological lithium problem' (abbreviated to CLP). Also, certain types of stars have an inexplicably large amount of Lithium. One idea has it that this could come from a novae (which should not be confused with a supernovae). A novae can occur when a White Dwarf is orbitted by a much larger star. Gas emitted by that star falls onto the White Dwarf causing an explosion. Claim to fame: Lithium is the lightest metallic element.
|
![]()