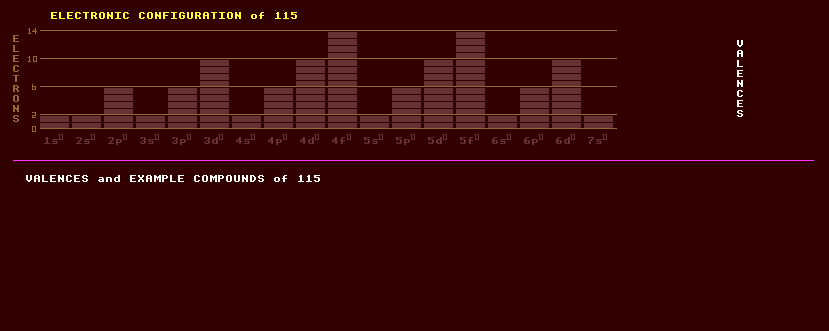

|
115
In February 2004 four atoms of Element 115 have been created by firing a beam of calcium-48 ions at a target of americium-243. It seemed to have a fairly long lifetime of about 90 milliseconds suggesting it is near to the predicted 'island of stability'. Two isotopes of element 115 were produced, 115-288 when 3 neutrons were emitted from the interaction of Am-243 with Ca-48, and 115-287 when 4 neutrons were emitted instead.
95Am243 + 20Ca48 → 115UUP287 + 40n1 The four atoms were observed to decay first to element 113 by alpha decay, and thence by further alpha decays to elements 115, 113 and 111 and finally by 3 more alpha decays to element-105, Dubnium. One of the nuclei took over a second to decay to element 111. Three of the decays ended up at Db-268, an isotope with a very long half-life (for that part of the Segre chart) of 16 hours. The fourth chain ended up at Db-267 when the energy of the colliding Ca-48 ions was slightly increased above the 248MeV used for the other three collisions. The whole alpha decay chain from element 115 to Dubnium took less than 30 seconds.
115Uup287 → 113Uut283 + 2He4 → ... Here are the four reactions observed: 287Uup115 → 283Uut113 + 4He2 (46.6 milliseconds) → 279Uuu111 + 4He2 (147 milliseconds) 288Uup115 → 284Uut113 + 4He2 (80.3 milliseconds) → 280uuu111 + 4He2 (376 milliseconds) 288Uup115 → 284Uut113 + 4He2 (18.6 milliseconds) → 279Uuu111 + 4He2 (1196 milliseconds) 288UUp115 → 284Uut113 + 4He2 (280 milliseconds) → 279Uuu111 + 4He2 (517 milliseconds) Claim to fame:
|
![]()