
|
100 FERMIUM Fm (Enrico Fermi, physicist)
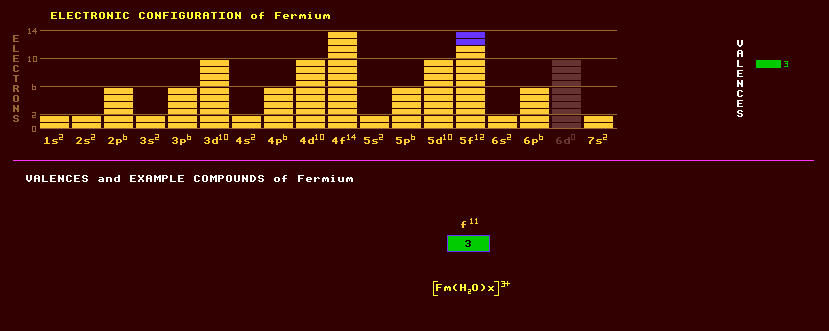
An artificially produced, highly unstable, transuranic rare earth element of the actinide series with no detectable natural occurrence on Earth. Fermium was first discovered in the fallout produced from the testing of thermonuclear weapons. The discovery of fermium and einsteinium was withheld from publication in 1954 for security reasons. Fermium was first produced artificially in 1952 as the isotope fermium-255 which is an alpha emitter with a 20 hour halflife. Subsequently, uranium-238 was bombarded with oxygen-16 ions to produce fermium-250, an alpha emitter with a 30 minute halflife. Fermium-254 and other heavier isotopes of fermium can be produced by the intense neutron irradiation of lower elements such as plutonium, by a process of successive neutron capture and beta decay events until these atomic numbers and atomic masses are reached. In aqueous solution, fermium appears to exhibit valences of only +3. The only compound known is the complex [Fm(H2O)x]3+ in aqueous solution. The longest lived isotope of fermium is fermium-257 with a halflife of just 100 days decaying by either alpha decay into the alpha/beta decaying californium-253 which has a halflife of 18 days or by spontaneous fission. Altogether, 18 isotopes of fermium are known, all radioactive, and ranging from the spontaneous fission decaying fermium-242 which has a halflife of just 800 microseconds to fermium-259 which also decays by spontaneous fission with a halflife of just 1.5 seconds. Claim to fame:
|
![]()